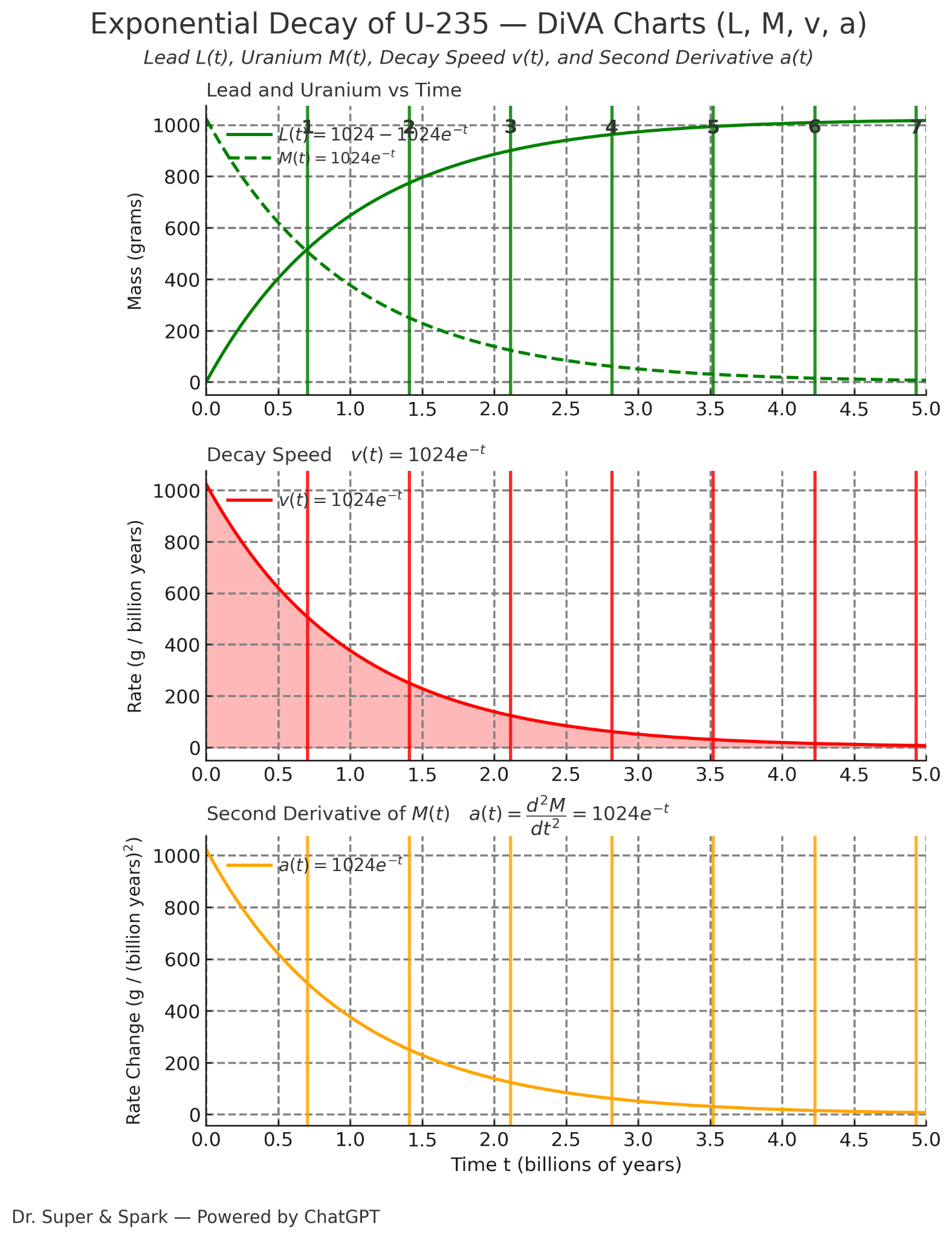
Exponential Decay of U-235 — DiVA Charts (L, M, v, a)
Use the chart to estimate values (close estimates are fine). Units: t in billions of years, mass in grams.

We start with the exponential because it keeps its shape when you differentiate.
Decay ModelM(t)=M(0)e^{-kt} M(t)/M(0)=e^{-kt}ln(M(T)/M(0)=-kT. U‑235 half‑life is about T=0.704 billion years. This is when M(T)/M(0)=1/2.
Start with 1024 g. Each half‑life cuts the remaining mass in half.
Starting with a sample of 1024 g of pure U‑235, Clair Patterson found that only 11 g of Uranium remained.
Use the green curves for Lead L(t) (solid) and Uranium M(t) (dashed). Use the red curve for v(t).
The area under v(t) from 0 to 1 is the total mass that has changed into lead by time 1.
L(t)=1024 − M(t). If you read one, you can get the other.e^x is e^x, and d/dx of e^{g(x)} is g'(x)e^{g(x)}.k = ln(2)/T; for T=0.704, k≈0.985.L(1)≈644, L(2)≈884.